-
Sign In
-

-
 Sony Biotechnology
Sony Biotechnology
-
-
 Sony Biotechnology
Sony Biotechnology
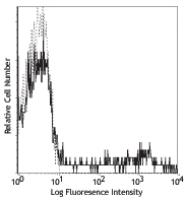
Purified anti-human HLA-DR
Antibodies Single
Sony
L243
Other
Mouse IgG2a, κ
Human
100 µg
2138010
$73.00
Description
HLA-DR is a heterodimeric cell surface glycoprotein comprised of a 36 kD α (heavy) chain and a 27 kD β (light) chain. It is expressed on B cells, activated T cells, monocytes/macrophages, dendritic cells, and other non-professional APCs. In conjunction with the CD3/TCR complex and CD4 molecules, HLA-DR is critical for efficient peptide presentation to CD4+ T cells.
Formulation
Phosphate-buffered solution, pH 7.2, containing 0.09% sodium azide.Recommended Usage
Each lot of this antibody is quality control tested by immunofluorescent staining with flow cytometric analysis. For flow cytometric staining, the suggested use of this reagent is ≤ 0.5 microg per 106 cells in 100 microL volume or 100 microL whole blood. It is recommended that the reagent be titrated for optimal performance for each application.
References
1. Brodsky F. 1984. Immunogenetics 19:179.
2. Robbins P, et al. 1987. Human Immunol. 18:301.
3. Stites D, et al. 1986. Clin. Immunol. Immunopathol. 38:161.
4. Warnke R, et al. 1980. J. Histochem. Cytochem. 28:771. (IHC)
5. Engleman E, et al. 1981. P. Natl. Acad. Sci. USA 78:1791. (IHC)
6. Zipf T, et al. 1981. Cancer Res. 41:4786.
7. Goodier M, et al. 2000. J. Immunol. 165:139. (Depletion)
8. Esser M, et al. 2001. J. Virol. 75:6173. (IP, WB)
9. Kalka-Moll WM, et al. 2002. J. Immunol. 169:6149. (Block)
10. Wang RF, et al. 1999. Science 284:1351. (Block)
11. Zaba LC, et al. 2007. J. Exp. Med. 204:3183. PubMed
12. Fujita H, et al. 2009. P. Natl. Acad. Sci. USA 106:21795. PubMed
13. Charles N, et al. 2010. Nat. Med. 16:701. (FC) PubMed
14. Goncalves RM, et al. 2010. Infect. Immun. 78:4763. PubMed
15. Yoshino N, et al. 2000. Exp. Anim. (Tokyo) 49:97. (FC)
16. Kim WK, et al. 2006. Am. J. Pathol. 168:822. (FC)
17. Stein R, et al. 2011. Leuk. Lymphoma 52:273.
18. Galkowska H, et al. 1996. Vet. Immunol. Immunopathol. 53:329.
19. Moro M, et al. 2005. BMC Immunol. 6:24.
20. Lauterbach N, et al. 2014. Mol Immunol. 59:19. PubMed